What is Distillation? Purpose, Types and Various Examples of Distillation
Distillation is an essential physical process of separation, but not a chemical reaction that is variedly used in chemistry, industry, and food science. Evidence shows that since at least 3000 BC in the Indus Valley Civilization, humans have been practicing distillation techniques using the terracotta distillation apparatus. Initially, it had been used to make perfumes, while the distillation of beverages occurred much later. In this article, you will get to know about the definition of distillation, its purpose, types, various examples, and are discussed below.
Distillation means a procedure by which two liquids having different boiling points can be separated. It is the boiling and subsequent condensation of a component selectively in a liquid mixture. The process of distillation starts with heating a liquid to the boiling point. The liquid evaporates and forms a vapor, and the gas is then cooled by passing it through pipes or tubes at a lower temperature. The cooled vapor then condenses, forming a distillate. The distillate is a purified form of the original liquid because when the liquid evaporates, impurities are left behind, and the distillate gets created without those impurities.
Repeating the process on the collected liquid is called double distillation, which improves the purity of the product. Mostly it is applied to liquids; however, the reverse process can also be used to separate gases by liquefying the components by making changes in temperature or pressure.
Raoult’s Law and Dalton’s Law
The distillation process is dependent on the two laws, and they are Dalton’s Law and Raoult’s Law for a mixture of liquids. According to Dalton’s law of partial pressures, the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of all the constituent gases. Raoult’s law, on the other hand, states that the partial pressure of a single liquid component in an ideal liquid mixture equals the product of the vapor pressure of the pure component and its mole fraction.
It is to be noted that the boiling point of the liquid depends on the surrounding pressure and changes accordingly. For instance, the boiling point of water at sea level is 100 degrees Celcius. However, its boiling point at an altitude of 1905 meters is 93.4 degrees Celsius as the pressure of the atmosphere is relatively lower at high altitudes.
A plant performing the process of distillation is called a distillery. When the distillation is performed in a laboratory, it uses batches of the liquid mixture, whereas, in industrial distillation processes, which are generally continuous, a constant composition of the mixture requires to be maintained.
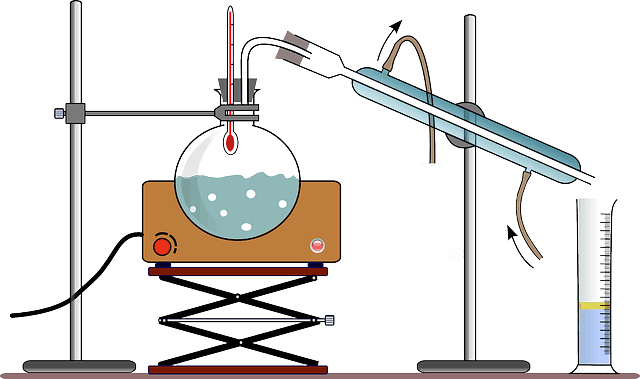
The apparatus that are used to perform distillation is called a still. The three most significant apparatus needed for distillation include the distillation flask that helps in heating up the mixture and volatilizes the components, the condenser that cools down the vapors back to a liquid state, and the collection vessel. Other requirements are a source of heat, fractioning column, thermometer, and Standard Glassware.
Purpose of Distillation
Distillation is used by chemists to purify compounds in solution or separating mixtures of solutes. This process is also used for commercial purposes. The purpose includes collecting different volatile ‘fractions’ as they are produced. In this separation technique, either the concentration of a particular component is increased in the mixture or an almost pure component is obtained from the mixture.
However, it is impossible to get a completely pure sample of a component from a mixture by distillation as a component in the mixture cannot have zero partial pressure. Distillation is also applied on an industrial scale for purification of the liquid products obtained from chemical synthesis.
Types of Distillation
Different types of distillation include:
- Simple distillation
- Fractional distillation
- Steam distillation
- Vacuum distillation
- Air-sensitive vacuum distillation
- Short path distillation
- Zone distillation
Simple Distillation
The simple distillation process is applied when the boiling points of two liquids are significantly different from each other (a minimum difference of 25-degree centigrade) or to separate liquids from solids or non-volatile components. It involves heating the mixture of liquid to the boiling point and immediately condensing the vapors so formed. The condensed vapor is then collected. Raoult’s law governs the purity of the distillate or the purified liquid.
Fractional Distillation
In fractional distillation, liquids in the mixtures that have similar boiling points are often separated. Several vaporization-condensation steps are involved in fractional distillation, which takes place in a fractioning column. This process is also known as rectification. With every vaporization-condensation cycle, the purity of the distillate improves.
Steam Distillation
Steam distillation is used to separate the components in a mixture that are heat-sensitive. In the beginning, the steam is passed through the mixture to make it slightly heated and vaporize some portion of it. A high heat-transfer rate is seen without requiring high temperatures. The vapors so formed are then condensed to get the necessary distillate. This distillation process is used to obtain essential oils and herbal distillates from several aromatic flowers/herbs.
Vacuum Distillation
For separating liquids that have very high boiling points in mixtures, the Vacuum Distillation is ideal. To boil these compounds, they are not heated to high temperatures, but the pressure of the surroundings is lowered. Due to lower pressure, the component boils at lower temperatures.
Once the component’ vapor pressure is equal to the pressure of the surrounding, it is transformed into a vapor. These vapors are then condensed as well as collected as the distillate. The method of vacuum distillation is also useful to obtain highly purified samples of compounds that decompose at high temperatures.
Air-Sensitive Vacuum Distillation
The vacuum distillation process is usually carried out for compounds that are sensitive to air and readily react with it. However, once the process is complete, the vacuum must be replaced with an inert gas. This process is often referred to as air-sensitive vacuum distillation.
Short Path Distillation
The purification of a small quantity of a compound that is unstable at high temperatures is possible through short path distillation. This process is called a short path because the distillate travels a minimal distance before being collected, and this is done under lowered pressure levels. As the distance traveled by the distillate is very short, this method reduces the wastage along the walls of the apparatus.
Zone Distillation
The zone distillation process involves the partial melting of a substance, and the condensation of the vapors formed to get a pure distillate. This process is usually carried out in a long container with the help of a zone heater.
Various Examples of Distillation
1. The simplest and most common example of distillation is the steam from a kettle, which gets deposited as drops of distilled water on a cold surface.
2. This process is used in the separation of alcoholic liquors from fermented materials for purification of alcohol by separating liquids from non-volatile solids.
3. It is used to separate two or more liquids having different boiling points, as in the case of separation of gasoline, kerosene, and lubricating oil from crude oil in crude oil refining, enabling safe storage and transportation.
4. Making liquefied gases from the air. For example, nitrogen, oxygen, and argon are distilled from the air.
5. The desalination of seawater is done through distillation.
6. Perfumes and flavorings for food are obtained from herbs and plants by distillation.
7. Distilled water is used in lead-acid batteries and low-volume humidifiers.
8. Other industrial uses include the processing of chemical products such as formaldehyde and phenol.
References:
https://www.thoughtco.com/what-is-distillation-601964