Phosphorus Cycle: Definition, Steps and Interesting Facts
Phosphorous is a crucial nutrient for plants and animals. For instance, it forms an integral component of genes and also plays a significant role in the Adenosine Triphosphate (ATP) energy cycle. Without phosphorous, you wouldn’t be able to contract your muscles.
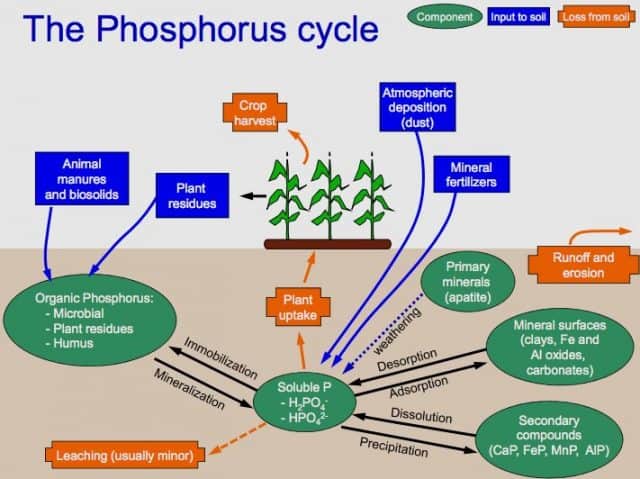
The phosphorus cycle refers to the biogeochemical cycle by which phosphorous moves through the biosphere, hydrosphere, and lithosphere. The atmosphere doesn’t play a substantial role in the cycling of phosphorous. This is because phosphorous and phosphorous-based compounds cannot be found in the air in the gas state.
They are normally solids at the standard pressure and temperature range found on Earth. So, phosphorus can mainly be found as tiny dust particles in the atmosphere. It mainly cycles through the soil, water, and sediments.
That means there is a land-based phosphorus cycle and water-based phosphorus cycle. Land-based cycle transfers phosphorus from soil to plants, to animals. It’s then cycled back to soil again. Water-based cycle, on the other hand, circulates phosphorous among the organisms living in the aquatic environment.
This article takes a detailed look at the process of the phosphorus cycle, including some important facts about the cycle. Let’s dive in.
Steps of Phosphorus Cycle
Phosphorus Cycle goes through 4 main steps:
- Weathering
- Absorption by Plants
- Absorption by Animals
- Return to the environment through decomposition
Shall we have a detailed look at the steps?
1. Weathering
In light of the fact that phosphorous is most commonly found in rocks, the phosphorus cycle starts in the Earth’s crust. Weathering causes phosphate salts to break from the rocks. The salts wash away into the ground, where they get mixed in the soil.
That takes us to the second step where phosphorous is taken up by plants and animals.
2. Absorption by Flora and Fauna
Plants absorb phosphate salts dissolved in soil water. It is worth noting that the quantities of phosphorus in the soil are normally small. This makes it one of the main factors that undermine plant growth.
That explains why we often apply phosphate fertilizers on agricultural land. Aquatic plants absorb inorganic form phosphorous from water and bottom layer of water bodies. In light of the fact that phosphate salts are not very water-soluble, they are also limiting factors for growth of plants in aquatic ecosystems.
3. Absorption By Animals
Animals absorb phosphorous by eating plants or plant-eating animals.
Namely, both marine and terrestrial plants are consumed by herbivorous animals, birds, and fish. Consequently, the organic form of phosphorus is transferred to the next level of consumers. Hence, the carnivores obtain phosphorous by eating herbivores.
It is worth noting that the rate of the phosphorus cycle is much faster in plants and animals than it is in rocks and sediments.
4. Return of Phosphorous Back to The Ecosystem
When plants, animals, birds, and fish die, they are deposited in soil and water bodies. Under conducive environmental conditions, bacteria and other microbes decompose the dead organisms. During decomposition, the organic form of phosphorous is converted into an inorganic form, which is then recycled to the soil and water.
After that, the element will end up in rock formations or sediments, where it will remain for millions of years. In the end, phosphorous is released again to the soil through weathering and absorbed by the plants. And the phosphorus cycle starts over.
Phosphorous stays in a particular ecosystem unit for a longer period, but it eventually recycles back to the environment. Hence, the percentage composition of this element remains at a virtually constant value.
According to scientific studies, the phosphorus cycle is the slowest of all the biogeochemical cycles. If you want to know other interesting facts about this cycle, read on.
Interesting Facts About Phosphorus Cycle
Fact 1: Hennig Brand is credited with discovering the element phosphorus in 1669 in Germany.
Fact 2: When he discovered phosphorous, Brand became the first person to be recognized for discovering an element.
Fact 3: You can most likely find phosphorous underground in rocks and soils. And it is essential for plant growth.
Fact 4: The majority of mined phosphorous is utilized in the manufacture of artificial fertilizers.
Fact 5: When excessive amounts of phosphorus-containing fertilizer are washed into the water bodies, it can result in algae bloom or increased algae growth.
Fact 6: The phosphorus cycle takes place at a much slower rate compared to other biogeochemical cycles. This is attributed to fact that the processes that drive the phosphorous occur at a slower rate.
Fact 7: Phosphorous plays a critical role in holding DNA (Deoxyribonucleic acid) together.
Fact 8: Phosphorous normally exists in a molecule combined with oxygen element.
Fact 9: Animals obtain phosphorous directly by consuming plants or indirectly by consuming plant-eating animals.
Fact 10: When phosphorous becomes part of the underwater sediment after being released from marine animals, it might remain there permanently.
Fact 11: Approximately 85% of the phosphorous present in the body of vertebrates, like us humans, can be found in the bones and teeth.
Fact 12: The availability of phosphorous in an environment is limited by the rate of its release during weathering.
Fact 13: Apatite [Ca5(PO4)3OH] is the main mineral with significant phosphorous content. And the production of phosphorous from its dissolution has a great influence on the productivity of an ecosystem.
Fact 14: The availability of organic phosphorous to promote plant, animal, and microbial growth depend on their degradation rate to produce free phosphate. There are several enzymes involved in the degradation. These enzymes include phytase, nucleases, and phosphatases.
Fact 15: Phosphine is the gaseous form of phosphorous. This gas is normally only found under laboratory conditions as Hydrogen phosphide (PH3). While phosphine is totally odorless in its pure form, it has a potent rancid garlic or fish smell in its impure disphosphane This gas is also highly flammable and toxic – a concentration of one part per million can quickly cause various short-term symptoms such as breathing problems and vomiting. Higher concentrations can be fatal.
Fact 16: Humans have caused significant changes to the global phosphorus cycle through various activities such as misuse of phosphorous fertilizers, shipping of phosphorus minerals, as well as shipping of food from farms to urban settings where it is lost as effluent. This has led to increased levels of phosphorous as pollutants in water bodies, resulting in eutrophication. Eutrophication has devastating effects on water ecosystems as it induces anoxic conditions.






